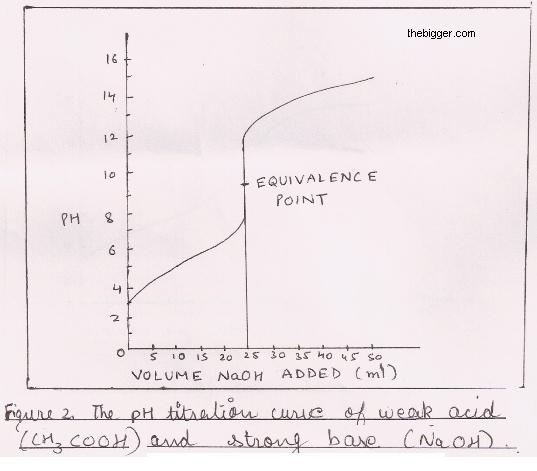
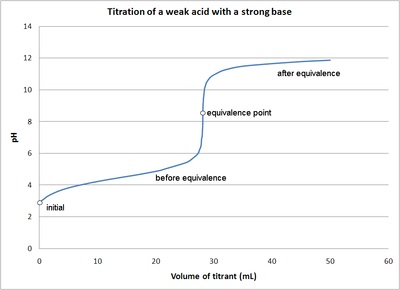
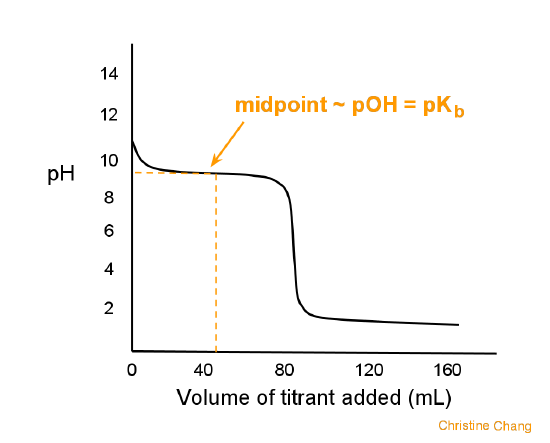
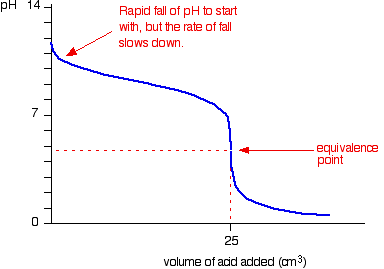
9. Investigate how pH changes for titrations of: weak acid vs. strong base | Secondary Science 4 All

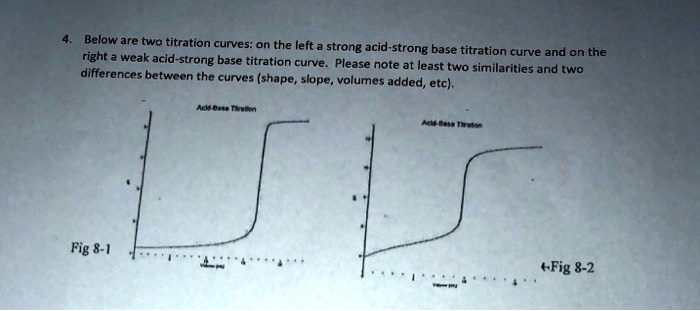
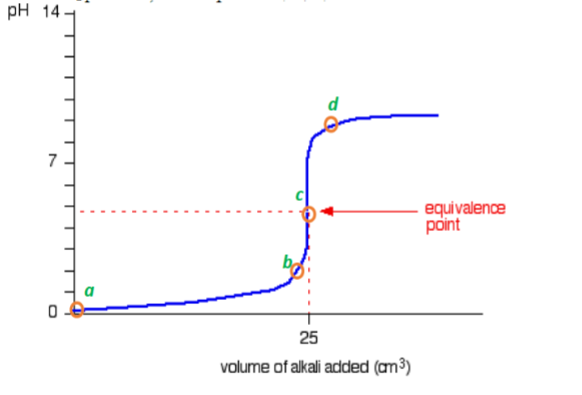
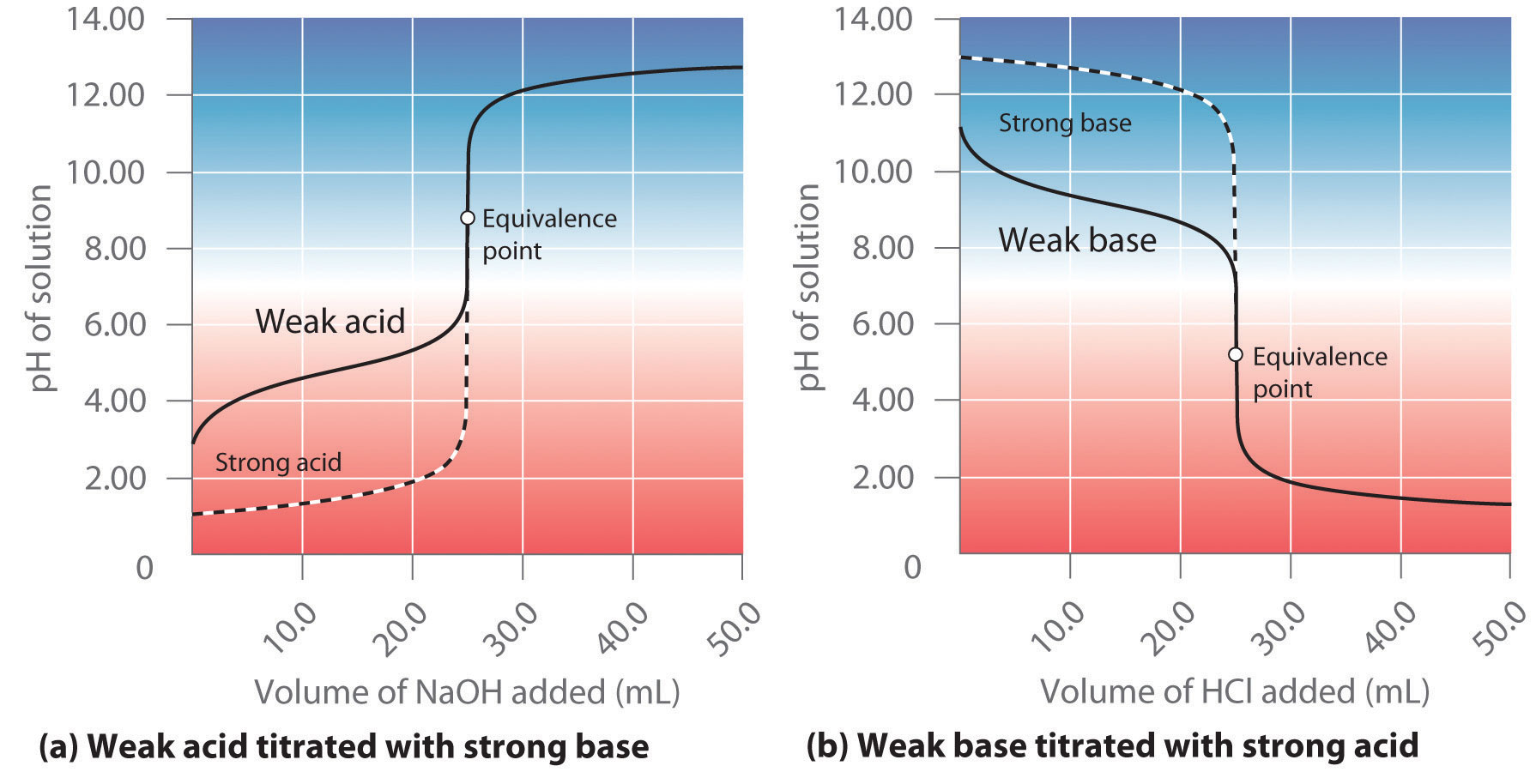
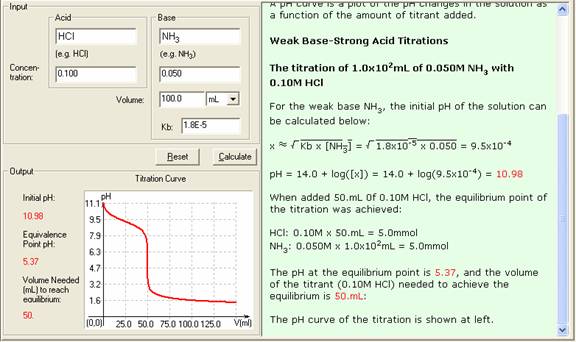
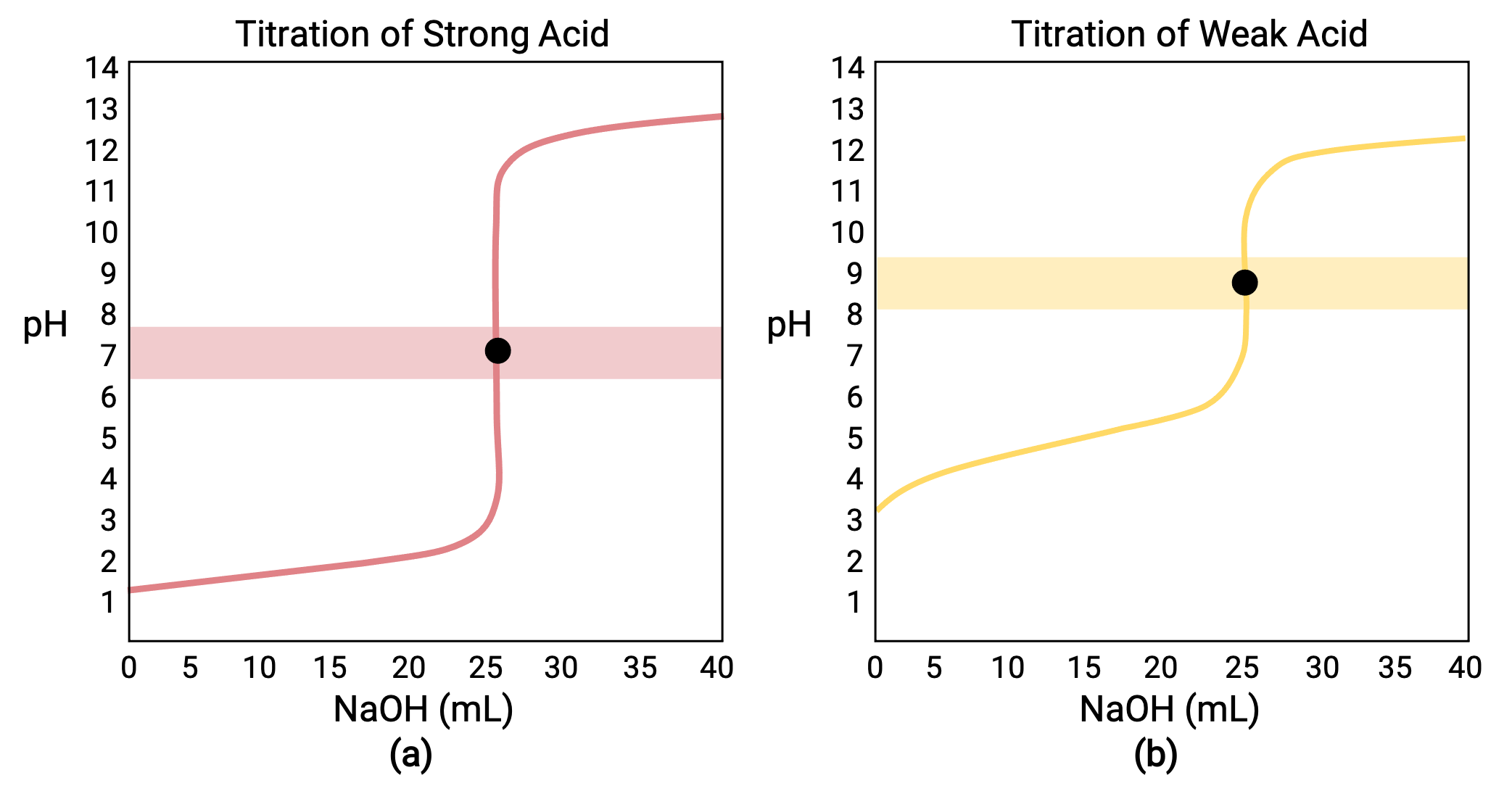
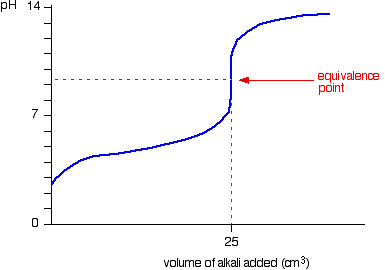
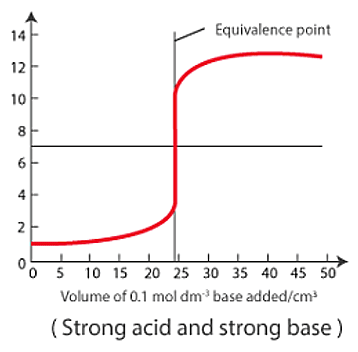
Here is an example of a titration curve, produced when a strong base is added to a strong acid. This curve shows how pH varies as 0.100 M NaOH is added to 50.0 mL of 0.100 M HCl.