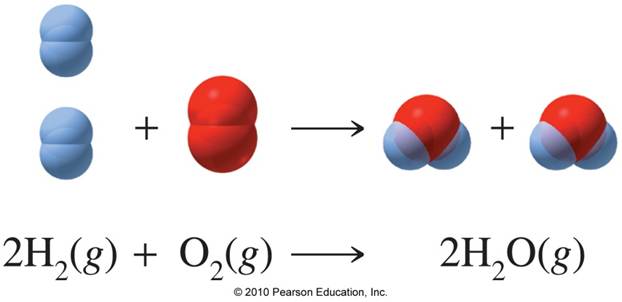
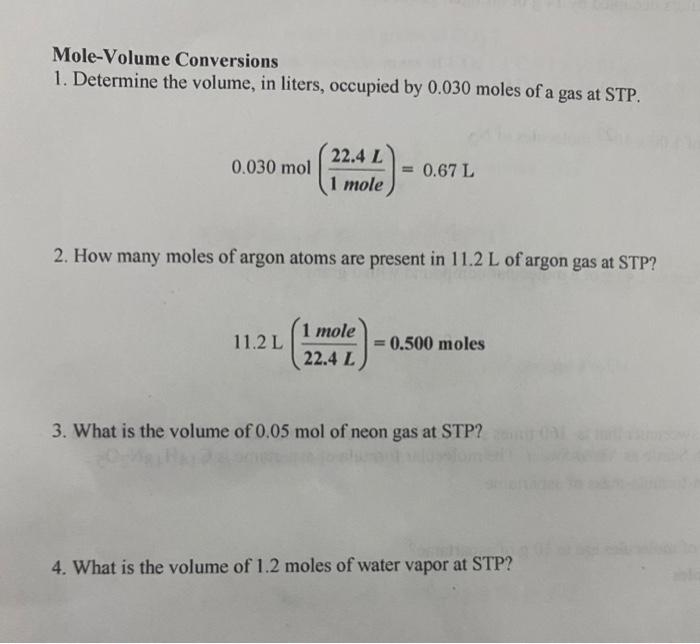SOLVED: Mole-Volume Conversions Determine the volume in liters occupied by 0.030 moles of a gas at STP. How many moles of argon atoms are present in 11.2 L of argon gas at

MOLARITY A measurement of the concentration of a solution Molarity (M) is equal to the moles of solute (n) per liter of solution M = n / V = mol / L Calculate. - ppt download

An equilibrium mixture in a vessel of capacity 100 litre contains 1 mol N2 , 2 mol O2 and 3 mol NO . No. of moles of O2 to be added so