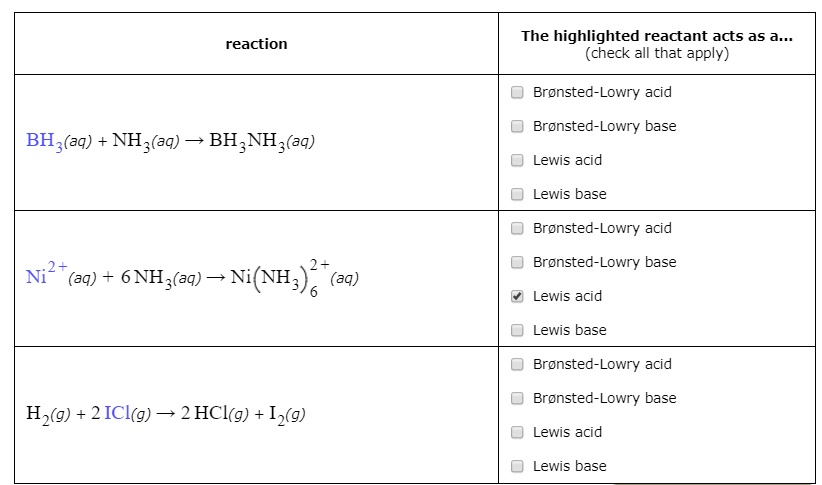
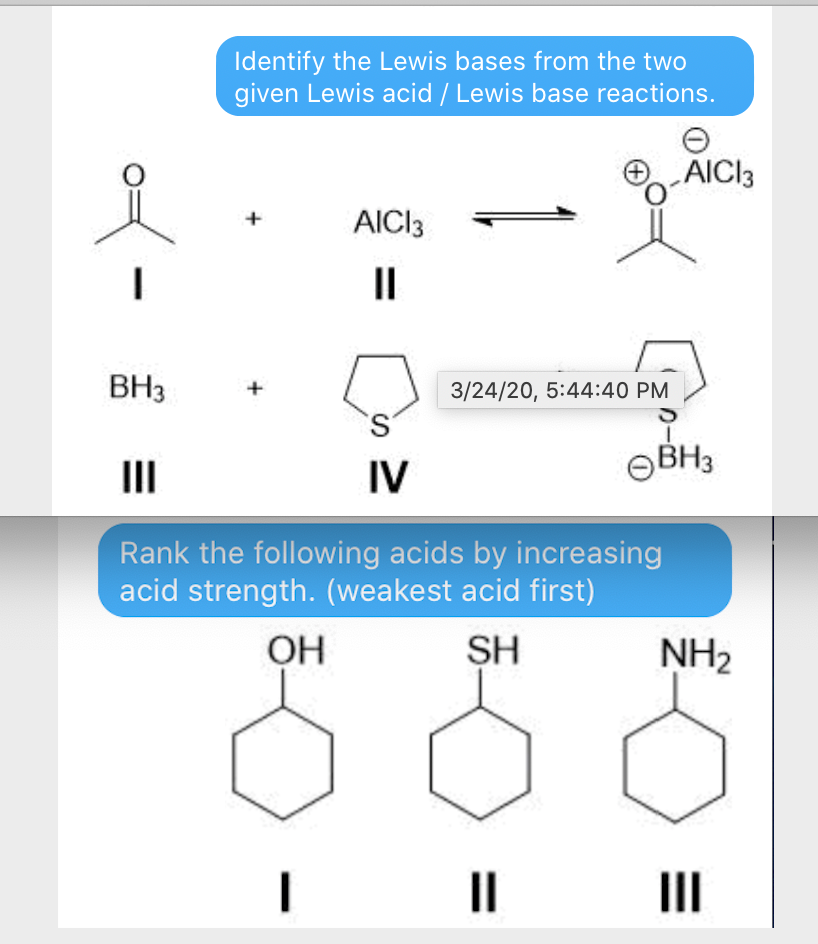
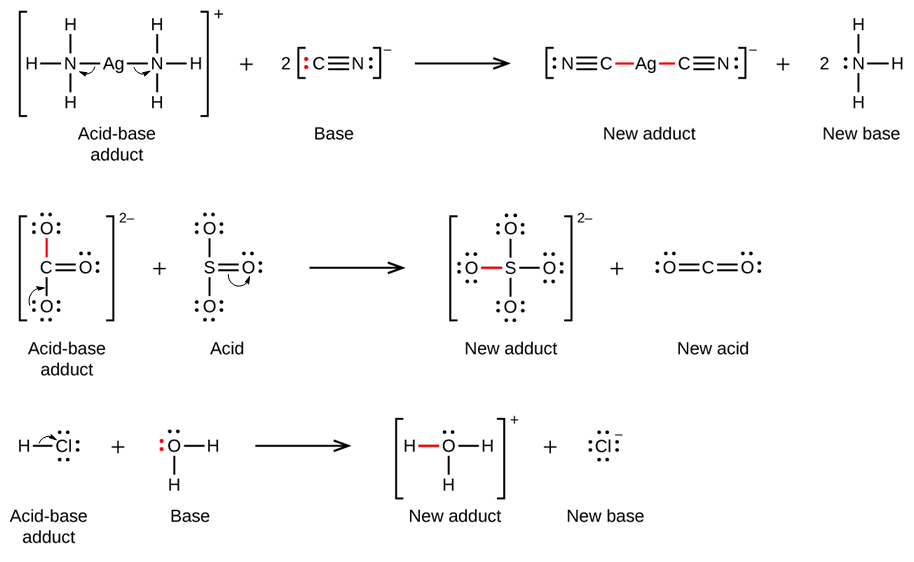
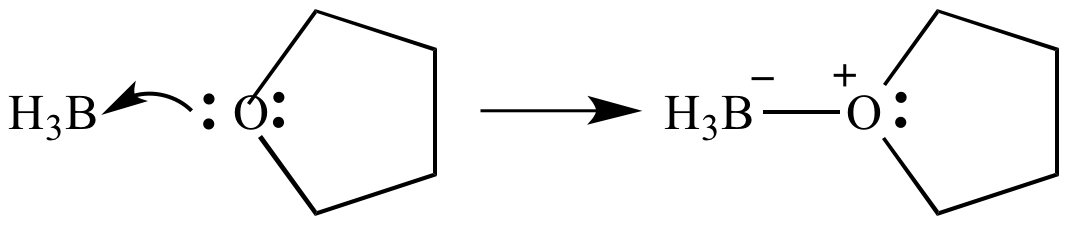
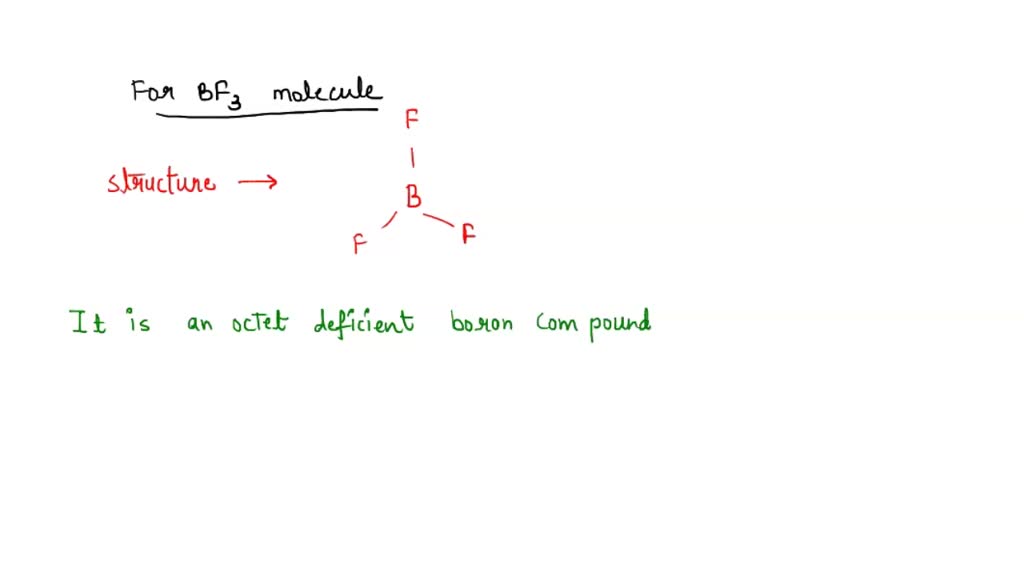
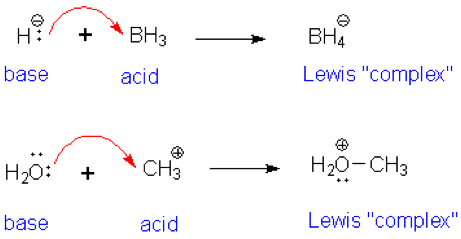
Nature and Strength of Lewis Acid/Base Interaction in Boron and Nitrogen Trihalides - Rodrigues Silva - 2020 - Chemistry – An Asian Journal - Wiley Online Library

When NH3 reacts with BH3, a boron-nitrogen bond is formed joining the two compounds. Which is the electrophile in this reaction? a. Electrophile BH3 b. Nucleophile NH3 | Homework.Study.com

Lewis acid-base adducts of zwitterionic alkali metal methanides and silanides with BH3 - ScienceDirect
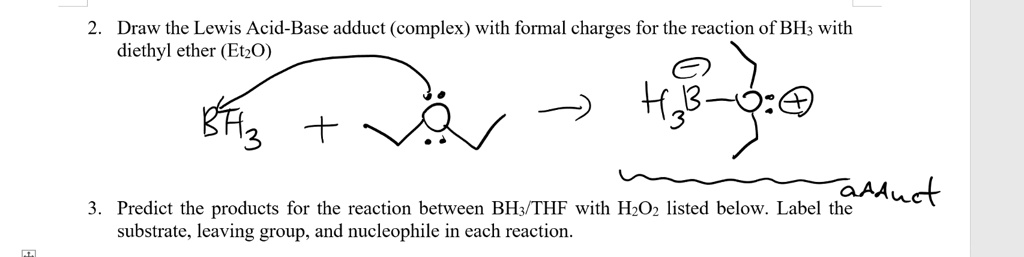
SOLVED: Draw the Lewis Acid-Base adduct (complex) with formal charges for the reaction of BH; with diethyl ether (EtzO) aAAuct Predict the products for the reaction between BH3 THF with HzOz listed