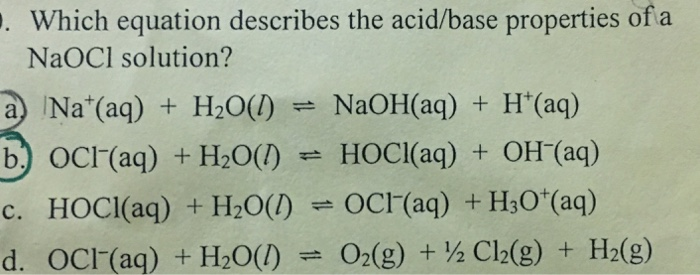
SOLVED: What is the dissolution reaction for solid NaClO?Once the ionic solid has dissolved, the anion that is formed is able to react as a base, with water as the acid. What

Lab 24 - Hydrolysis A salt formed between a strong acid and a weak base is an acid salt. Ammonia is a weak base, and its salt with any strong acid gives. -

HClO is a weak acid (Ka = 4.0 × 10–8) and so the salt NaClO acts as a weak base. What is the pH of a - Brainly.com

A). Acid-base titration of raw MWCNTs I=0.01M NaClO 4 and T=20 ºC. The... | Download Scientific Diagram
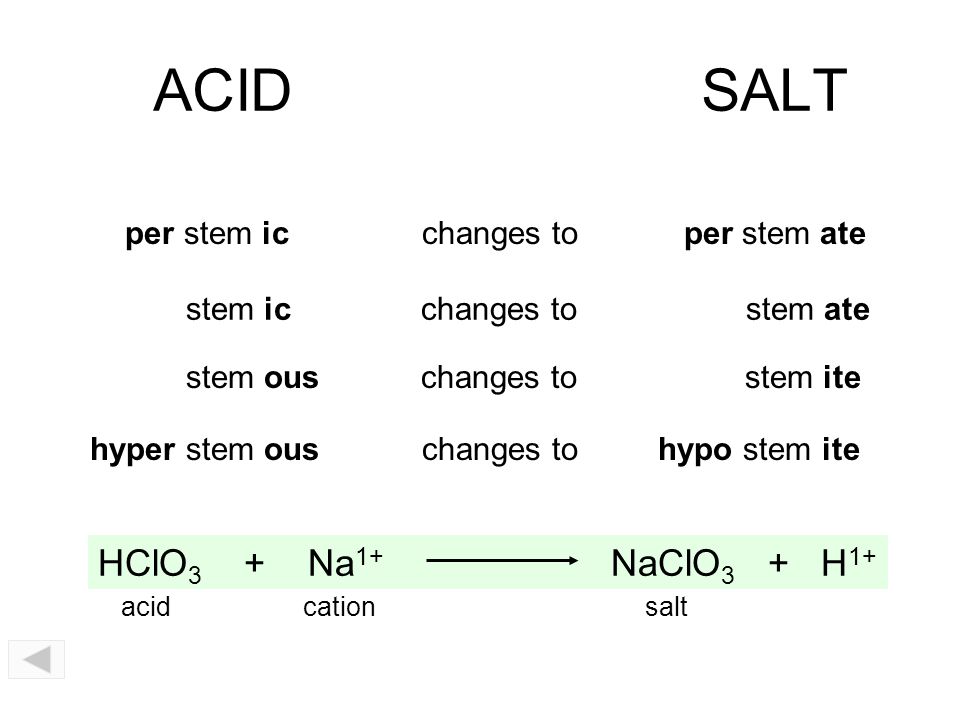
Acids, Bases, and Salts You should be able to Understand the acid-base theories of Arrhenius, Brønsted-Lowry, and Lewis. Identify strong acids and. - ppt download

HClO is a weak acid (Ka = 4.0 x 10-8) and so the salt NaClO acts as a weak base. What is the pH of a solution that is 0.034 M in
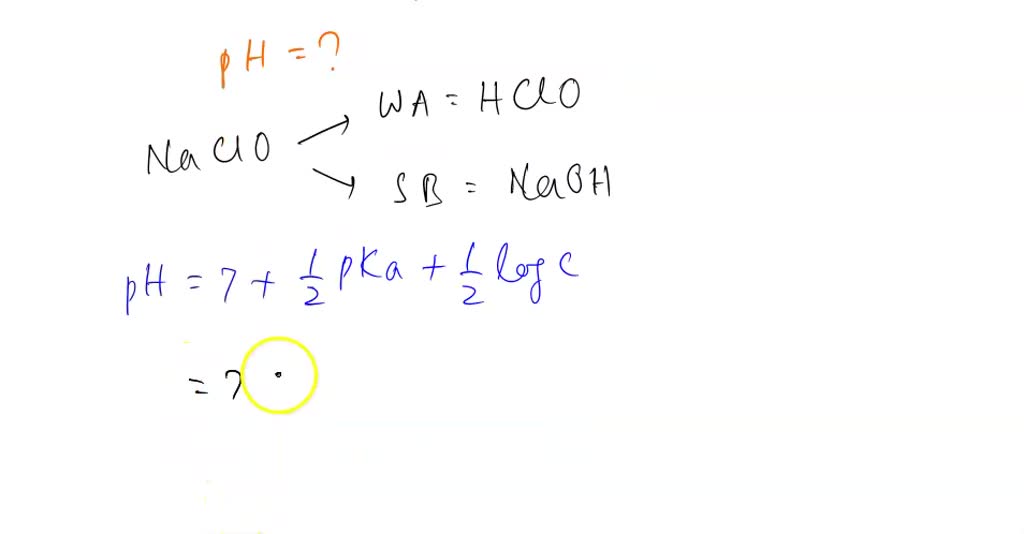










![47 Chemistry QPack] How do you know that HClO and NaClO are conjugates of each other? : r/Mcat 47 Chemistry QPack] How do you know that HClO and NaClO are conjugates of each other? : r/Mcat](https://preview.redd.it/8rsg2yxkum831.jpg?width=640&crop=smart&auto=webp&s=ab0ed8cf6296dab3cf389a686d1f90e42e9178b8)