Potassium Phosphate as a Solid Base Catalyst for the Catalytic Transfer Hydrogenation of Aldehydes and Ketones | ACS Catalysis

Variation of the nature of the base for the O-methylation of glycerol... | Download Scientific Diagram
CoII immobilized on an aminated magnetic metal–organic framework catalyzed C–N and C–S bond for

SOLVED: The balanced chemical equation for the reaction of the base potassium hydroxide with phosphoric acid to form potassium phosphate and water is: 3 KOH HPO4 KaPOs 3 HzO How many grams

K3PO4-Catalyzed Regiospecific Aminobromination of β-Nitrostyrene Derivatives with N-Bromoacetamide as Aminobrominating Agent | The Journal of Organic Chemistry

The potential of K3PO4, K2CO3, Na3PO4 and Na2CO3 as reusable alkaline catalysts for practical application in biodiesel production - ScienceDirect

EASTCHEM Food Grade 97+% Content(K3PO4,Dry Basis) Tripotassium Phosphate of,CAS NO.:7778-53-2(500g) : Amazon.sg: Industrial & Scientific

a) Evolution of the characteristic C=O IR band at 1815 cm -1 during the... | Download Scientific Diagram

What is the formula of a compound that contains K^+ and PO_4^{3-} ions? (a) K_3P (b) K_2PO_3 (c) KPO_4 (d) K_3PO_3 (e) K_3PO_4 | Homework.Study.com

A Regioselective Synthesis of 6-Alkyl- and 6-Aryluracils by Cs2CO3- or K3PO4-Promoted Dimerization of 3-Alkyl- and 3-Aryl-2-Propynamides. | Semantic Scholar

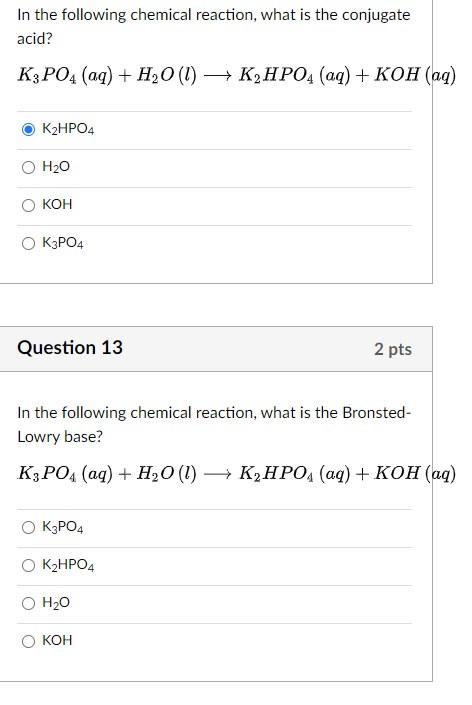

![ANSWERED] Classify the following as acid-base react... - Physical Chemistry ANSWERED] Classify the following as acid-base react... - Physical Chemistry](https://media.kunduz.com/media/sug-question/raw/52289625-1659182550.4991379.jpeg)