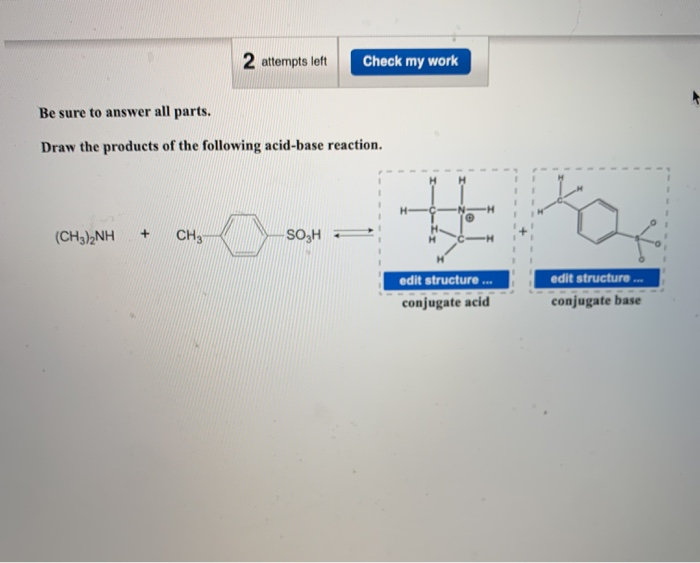
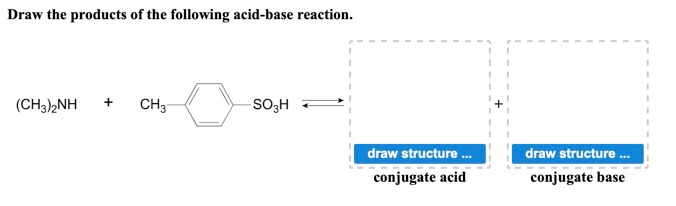
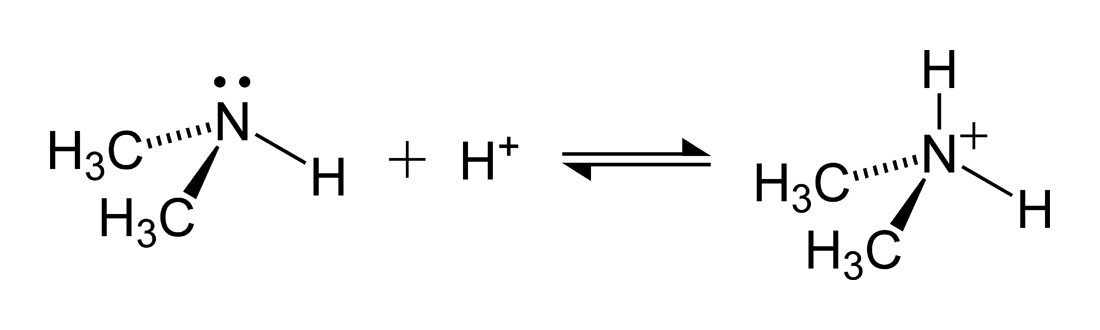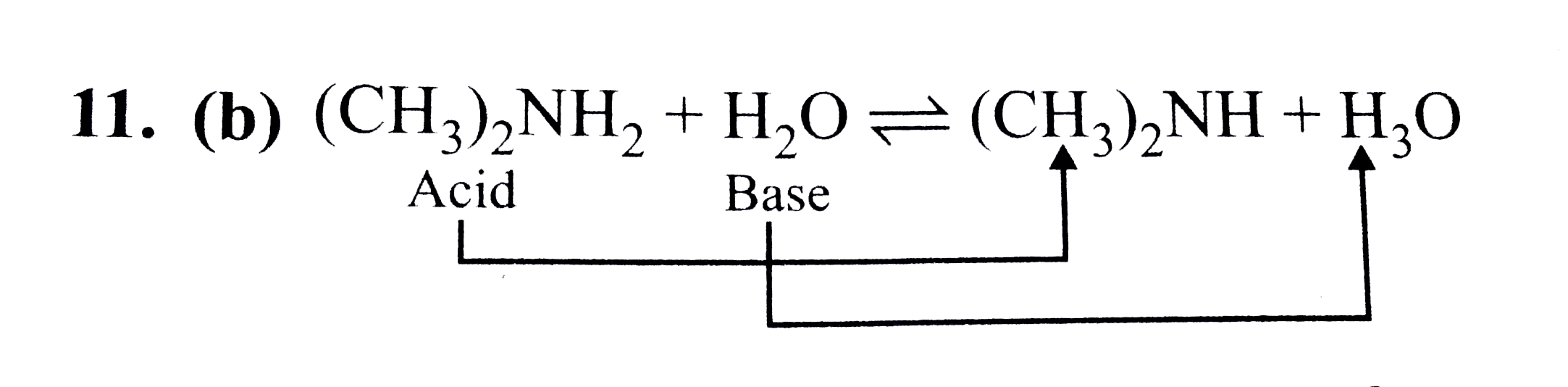Provide the structure of the conjugate acid and the conjugate base of dimethylamine ((CH3)2NH). | Homework.Study.com

Draw the product formed when the Lewis acid (CH3CH2)3C reacts with the Lewis base (CH3)2NH. - Brainly.com
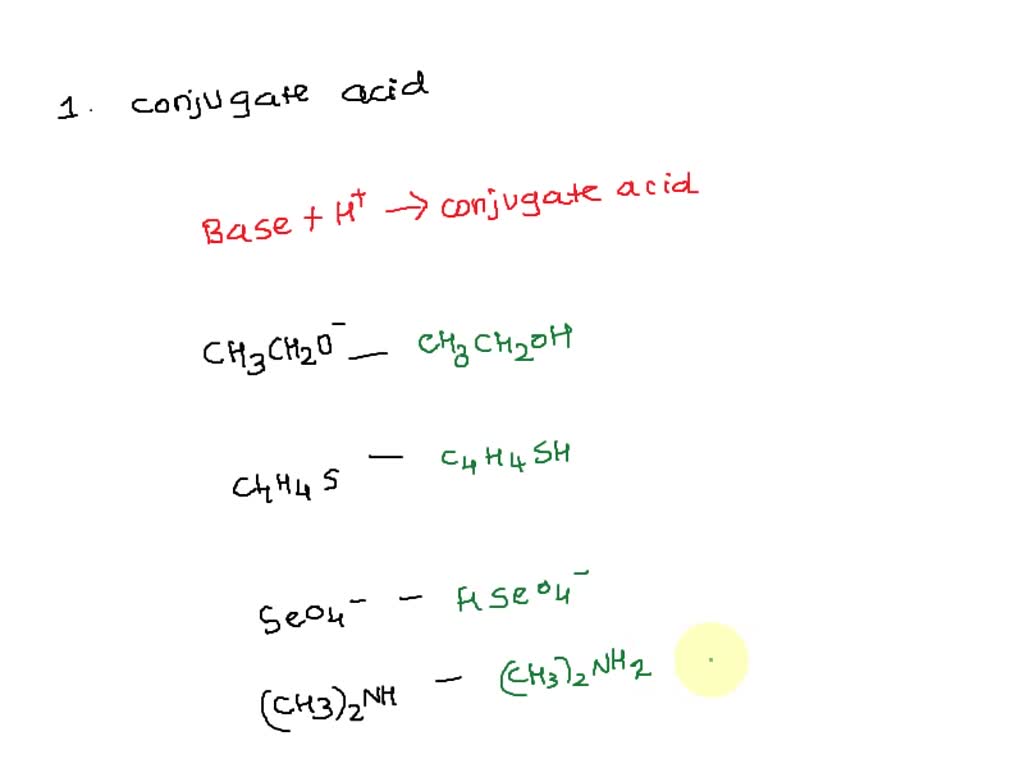
SOLVED: Q: 1). Write the formulas of the conjugate acids of the following Brønsted-Lowry bases. - CH3CH2O− - C4H4S - SeO-24 - (CH3)2NH 2). Enter the formulas of the conjugate bases of
Give reasons : (i) (CH3)2NH is more basic than (CH3)3N in an aqueous solution. - Sarthaks eConnect | Largest Online Education Community

Using the Brønsted-Lowry concept of acids and bases, which is the Brønsted-Lowry acid and base in the following reaction? (CH3)2NH(g)+BF3(g)→( CH3)2NHBF3(s) | Socratic

14.37 | Which base, CH3NH2 or (CH3)2NH, is the stronger base? Which conjugate acid, (CH3)2NH2+ or - YouTube