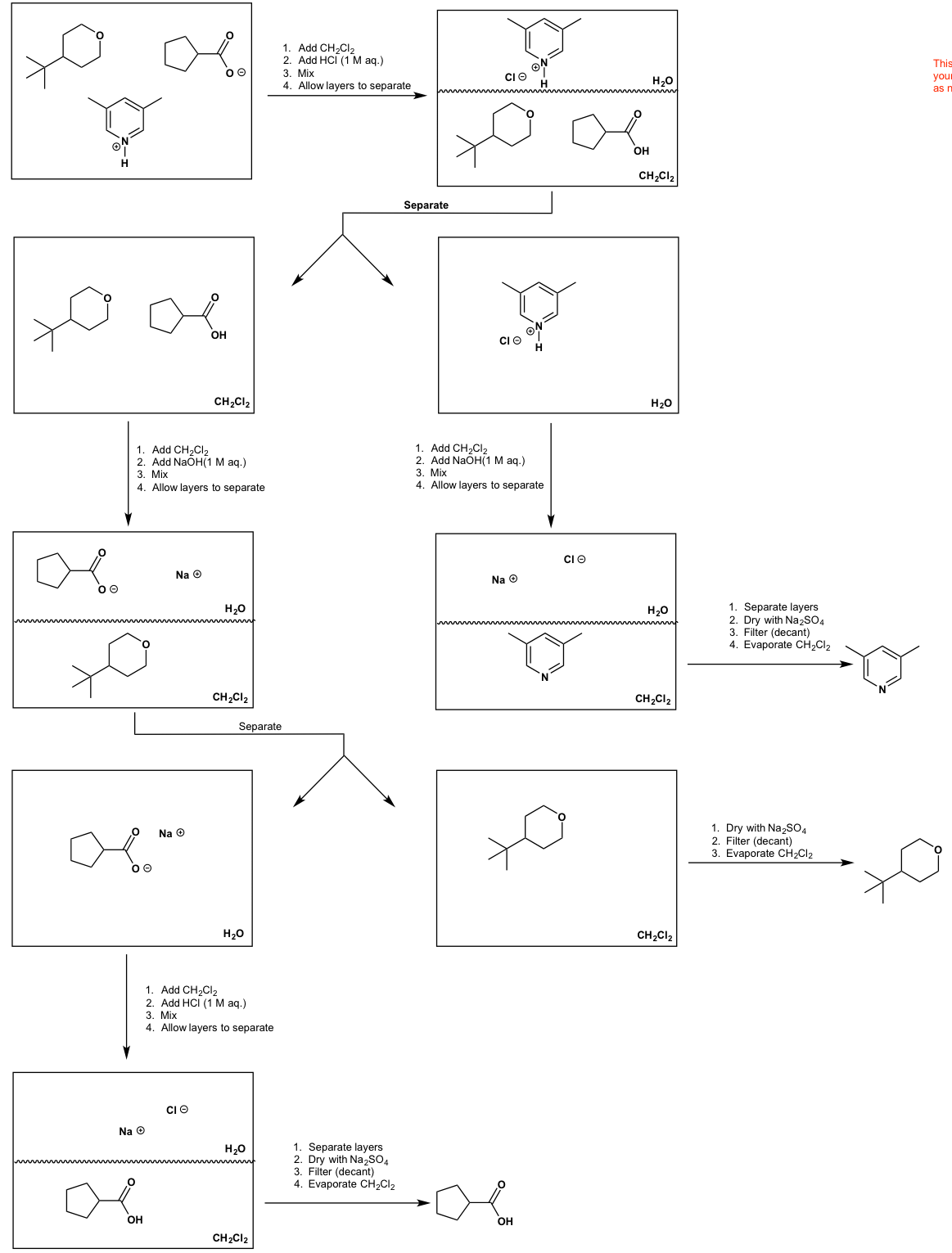
Base‐free Enantioselective C(1)‐Ammonium Enolate Catalysis Exploiting Aryloxides: A Synthetic and Mechanistic Study - McLaughlin - 2019 - Angewandte Chemie International Edition - Wiley Online Library

Acid-base properties of tetrapyrazinoporphyrazines. 1. Deprotonation of octaethyltetrapyrazinoporphyrazine in CH2Cl2, THF, DMSO and pyridine. The crucial role of water - ScienceDirect

Reaction of N,N-Dimethyltryptamine with Dichloromethane Under Common Experimental Conditions | ACS Omega

Which of the following would be extracted into the organic layer when using dichloromethane and sodium hydroxide as your extraction solvents? | Homework.Study.com

The Partition Coefficient Extraction of Benzoic Acid and its Conjugate Base Lab Report - The - Studocu
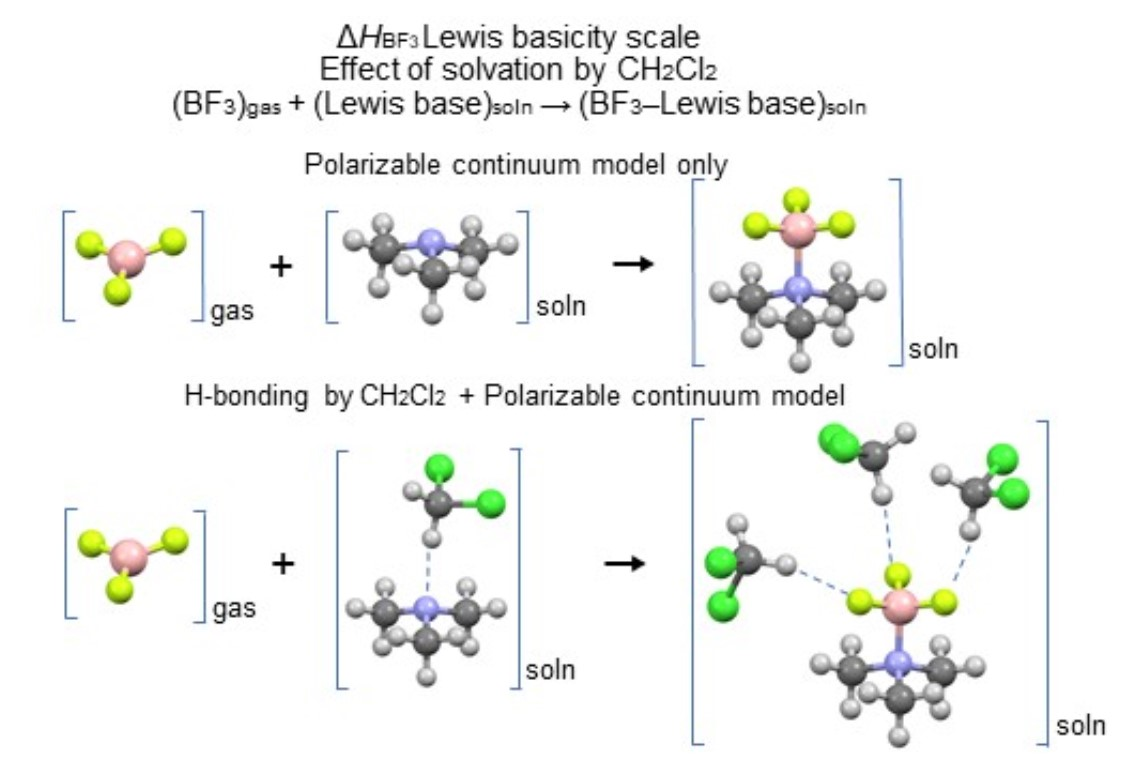
Molecules | Free Full-Text | Enthalpies of Adduct Formation between Boron Trifluoride and Selected Organic Bases in Solution: Toward an Accurate Theoretical Entry to Lewis Basicity
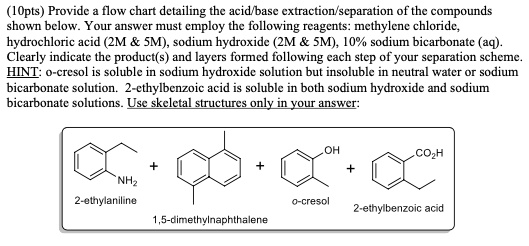
SOLVED: (1Opts) Provide flow chart detailing the acid base extraction scparation Of the compounds shown below. Your answer must employ the following reagents: methylene chloride; hydrochloric acid (ZM SM), sodium hydroxide (2M

A) Acid-and base-induced structural conversion between ins-SP-PPE and... | Download Scientific Diagram
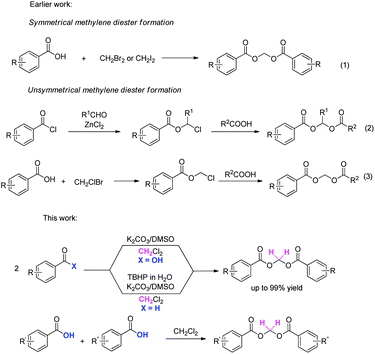


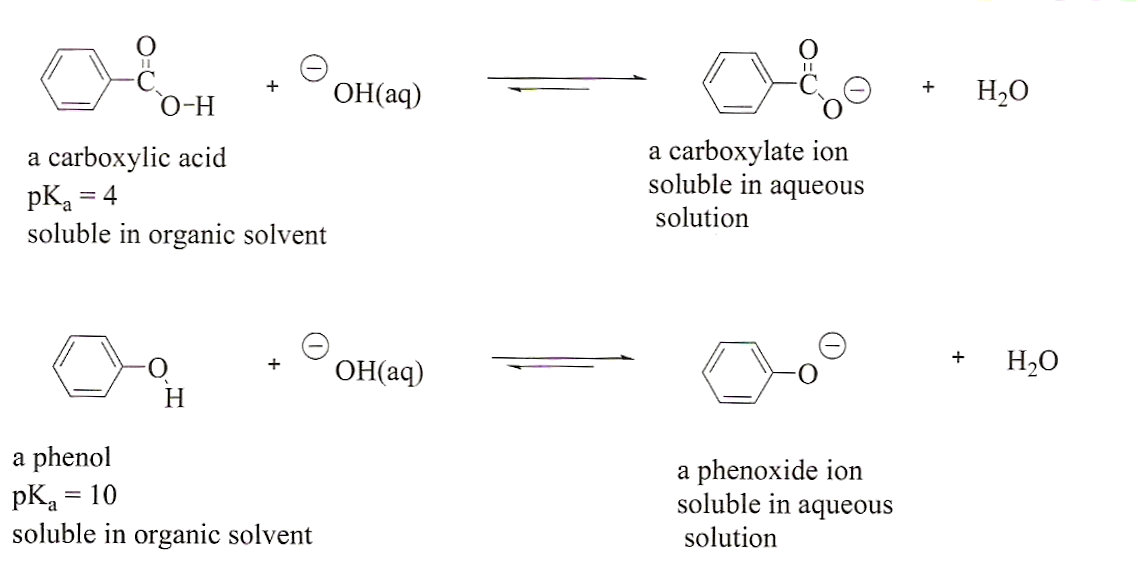
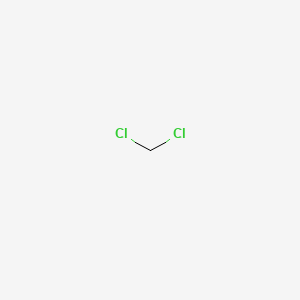
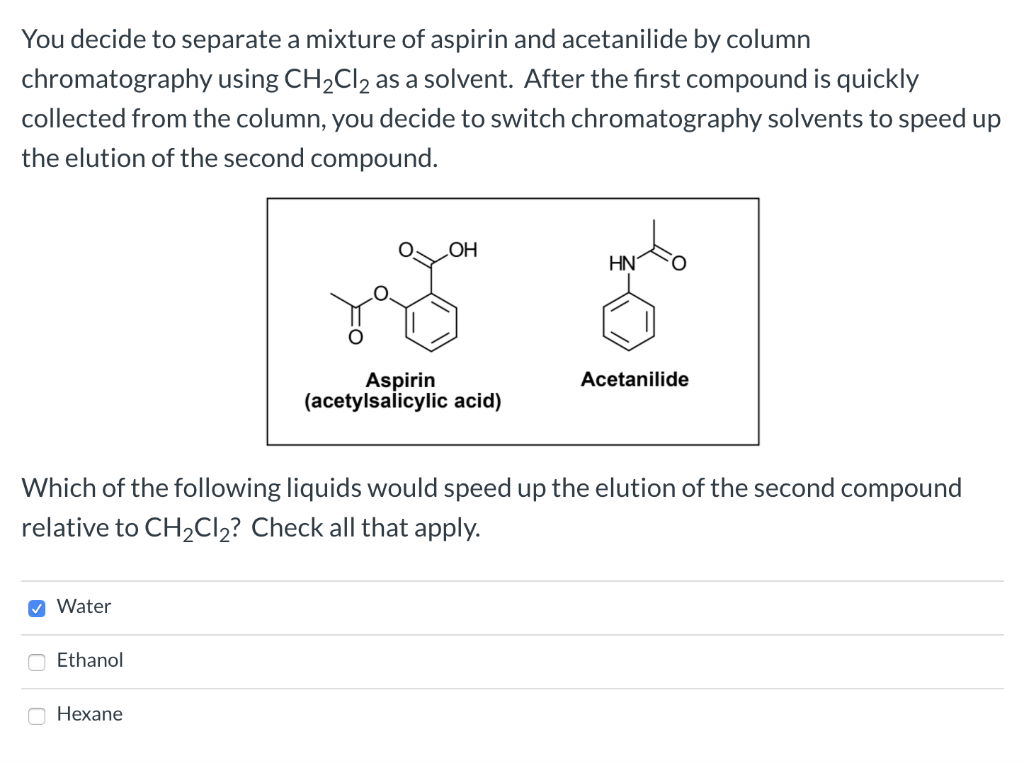
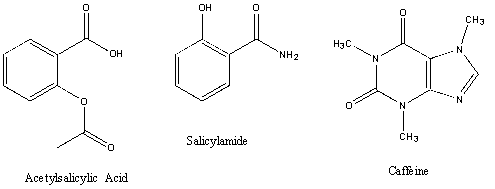
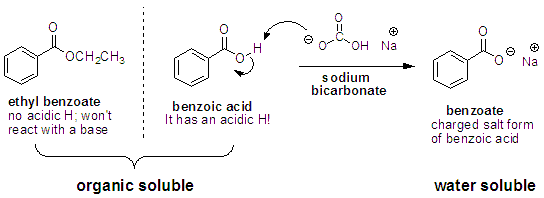



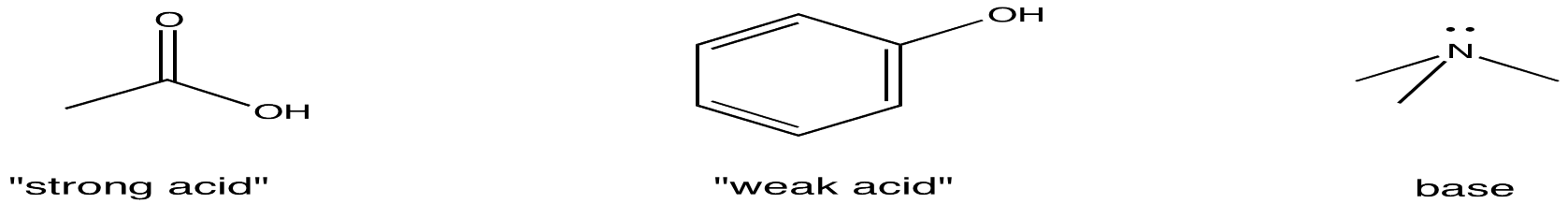
.jpg)