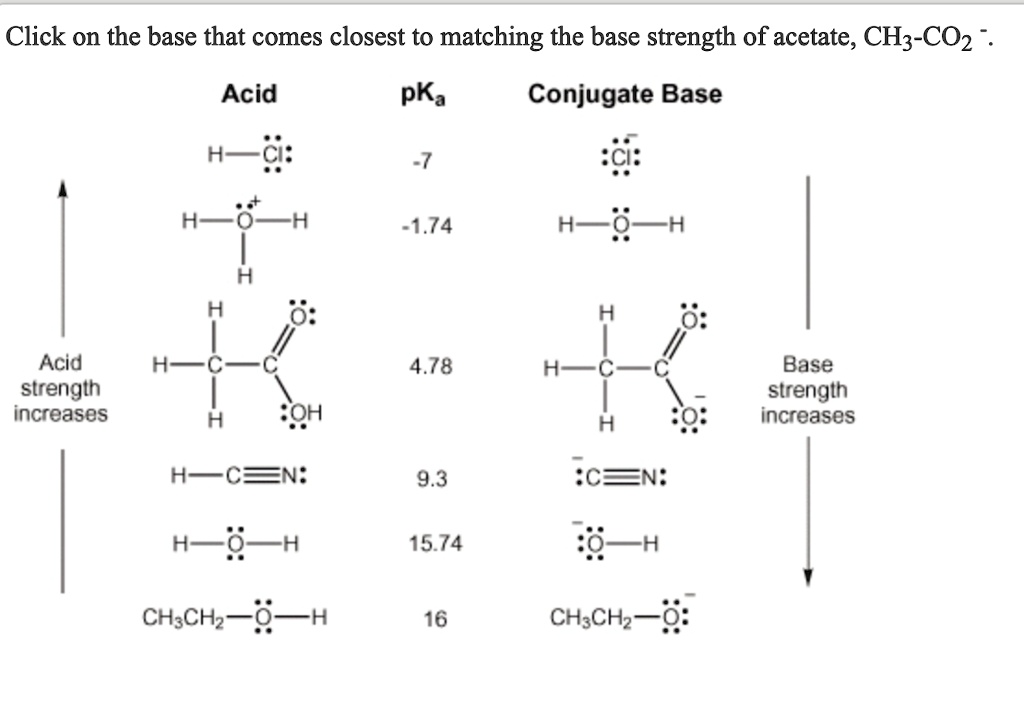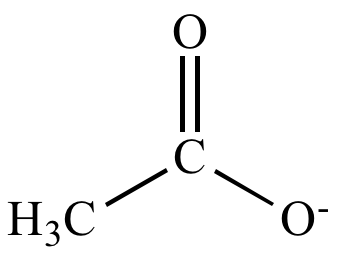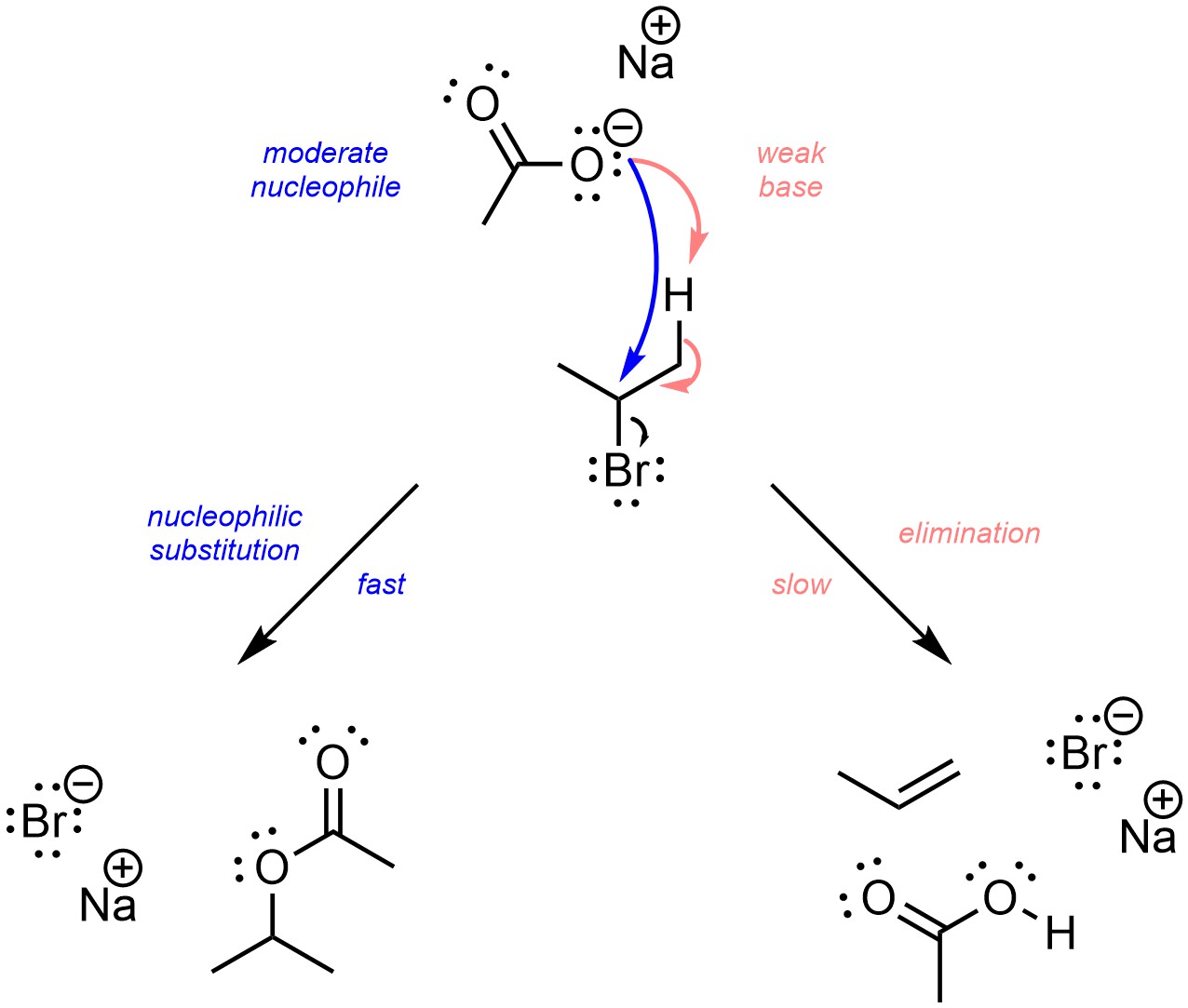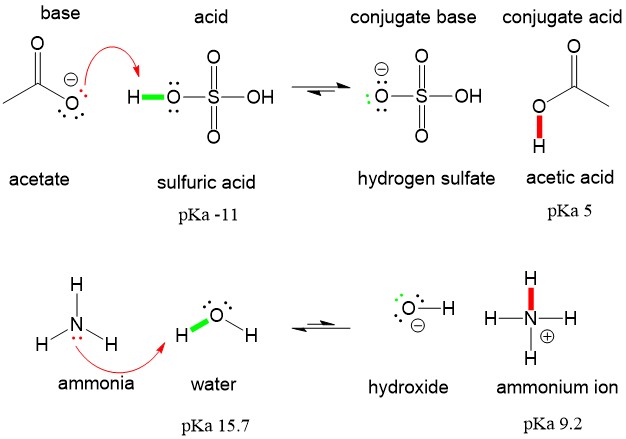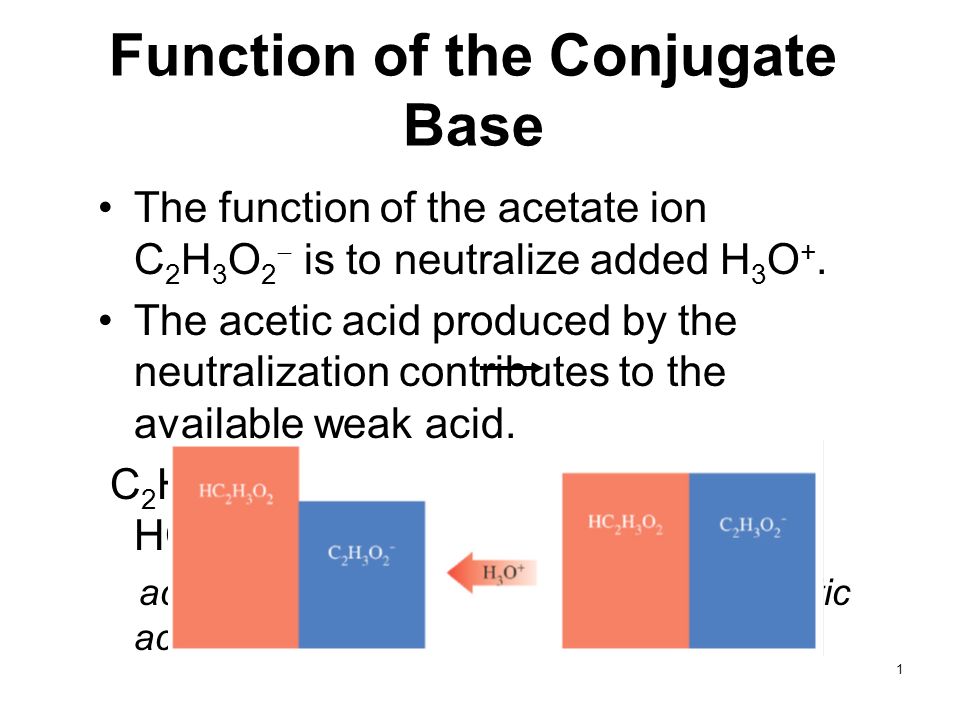
1 Function of the Conjugate Base The function of the acetate ion C 2 H 3 O 2 is to neutralize added H 3 O +. The acetic acid produced by the neutralization. - ppt download

Interplay of Acid–Base Ratio and Recycling on the Pretreatment Performance of the Protic Ionic Liquid Monoethanolammonium Acetate | ACS Sustainable Chemistry & Engineering

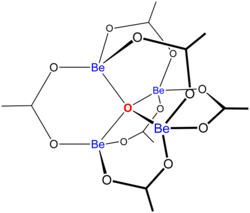
Basic beryllium acetate. II. The structure analysis - Tulinsky - 1959 - Acta Crystallographica - Wiley Online Library

3d Structure of Acetate, a Salt Formed by the Combination of Ace Stock Illustration - Illustration of biochemistry, acid: 84012279
Why does the solution of sodium acetate give more concentration of Hydroxide ion? Shouldn't the number of Hydroxide ion and hydrogen ion be equal? - Quora

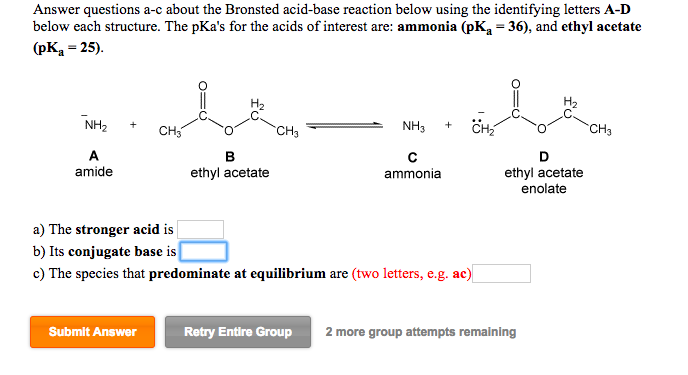
Can methyl acetate undergo a self-condensation reaction in the presence of an appropriate base? Explain. | Homework.Study.com

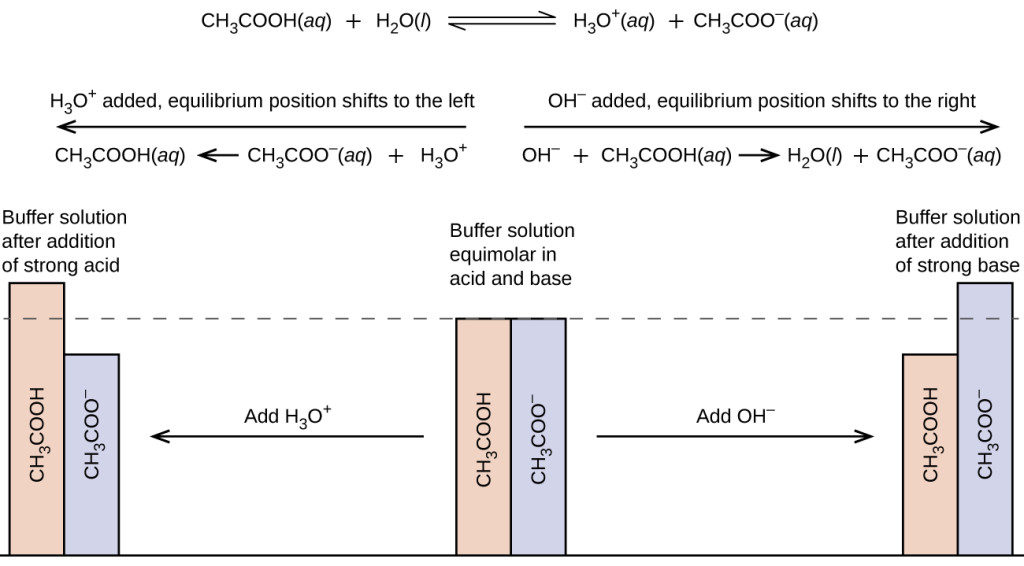
SOLVED: For conjugate acid-base pairs, how are Ka and Kb related? Consider the reaction of acetic acid in water CH3CO2H(a q)+H2O(I) ⇌CH3CO2^-(a q)+H3O^+(a q) where Ka=1.8 × 10^-5 a. Which two bases

Acétate d'indium(III), 99,99 %, (base métallique à l'état de traces), anhydre, Thermo Scientific Chemicals | Fisher Scientific